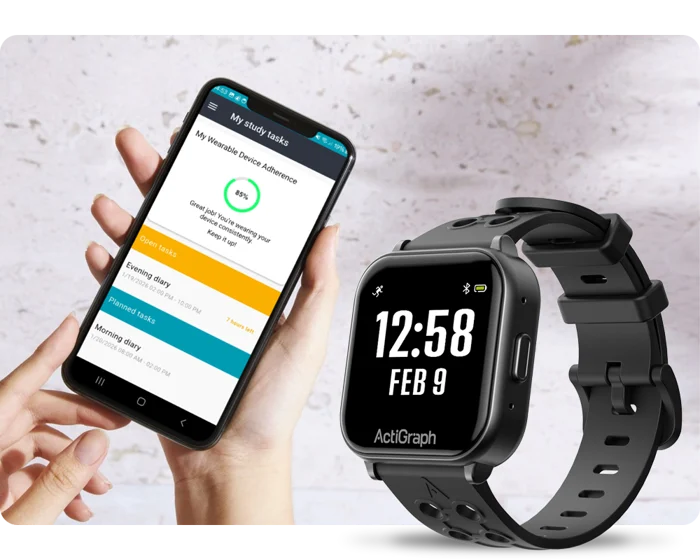
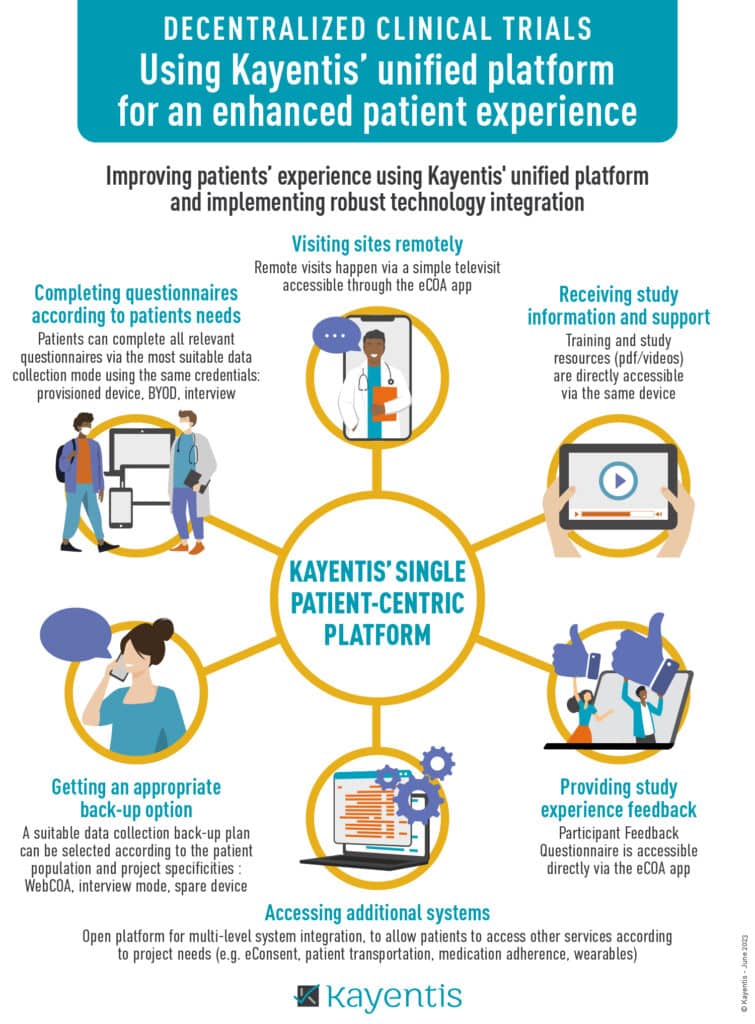
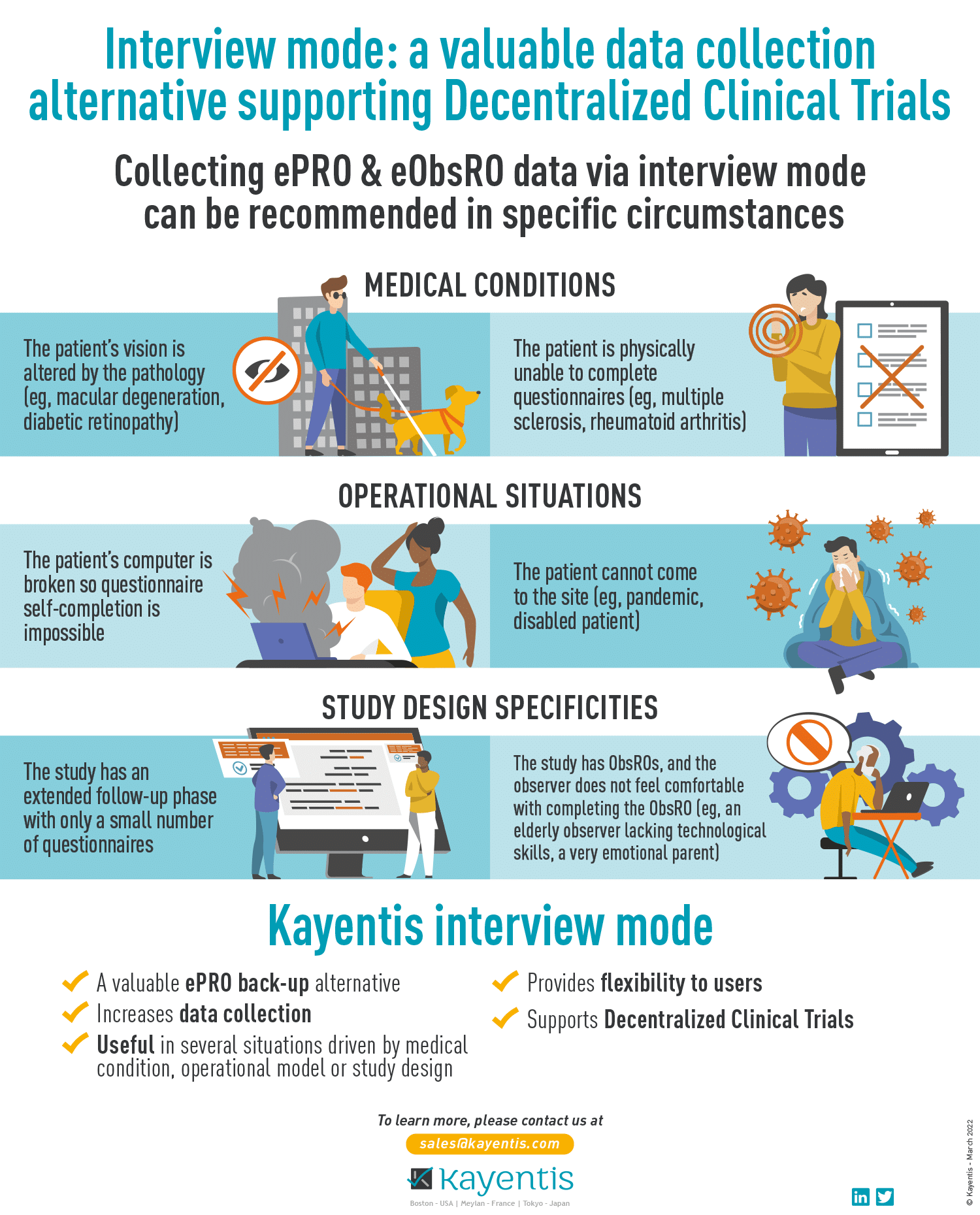
Press Release
Kayentis and Perceptive eClinical partner to tackle increasing digital system complexity in global clinical trials
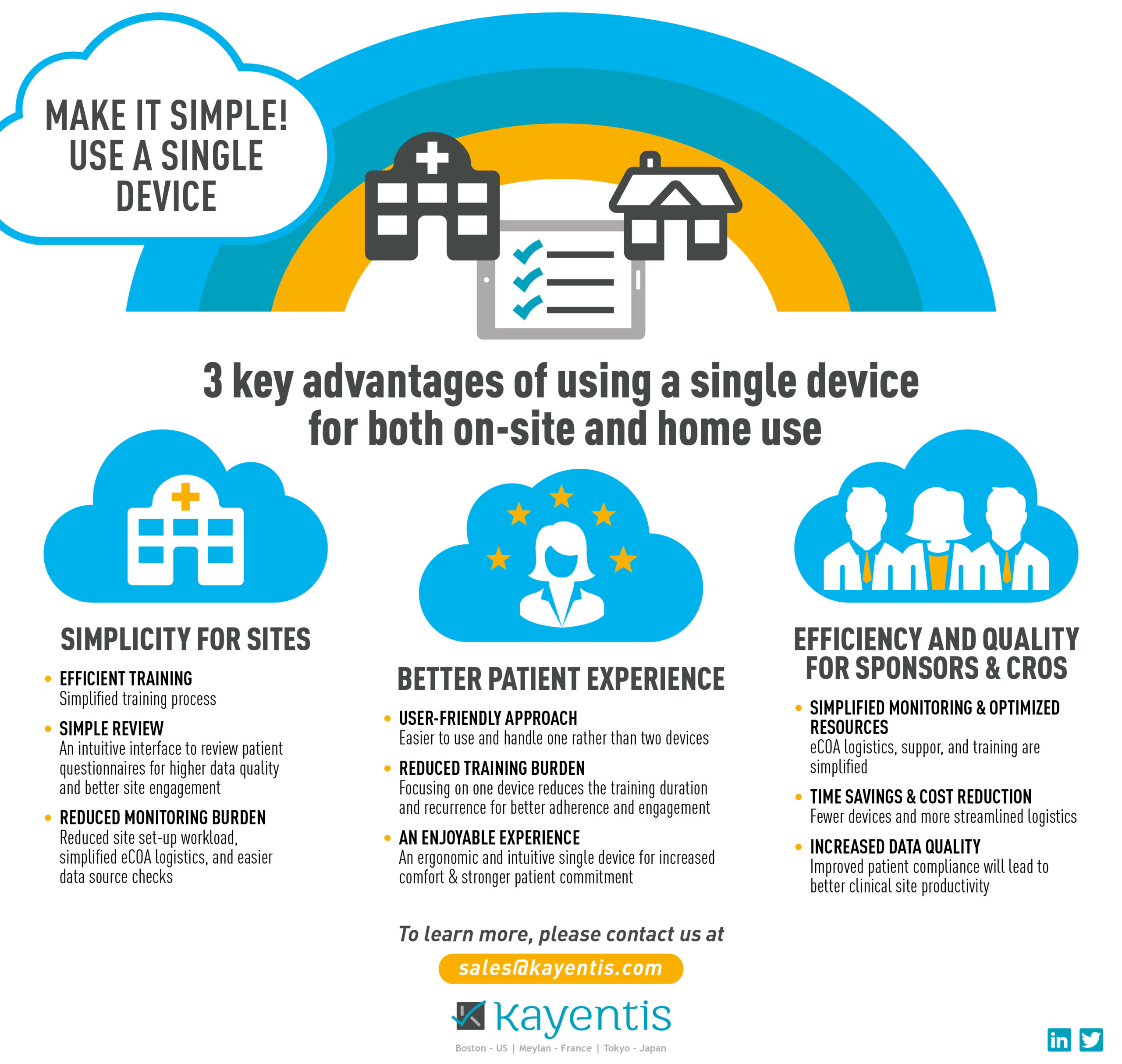
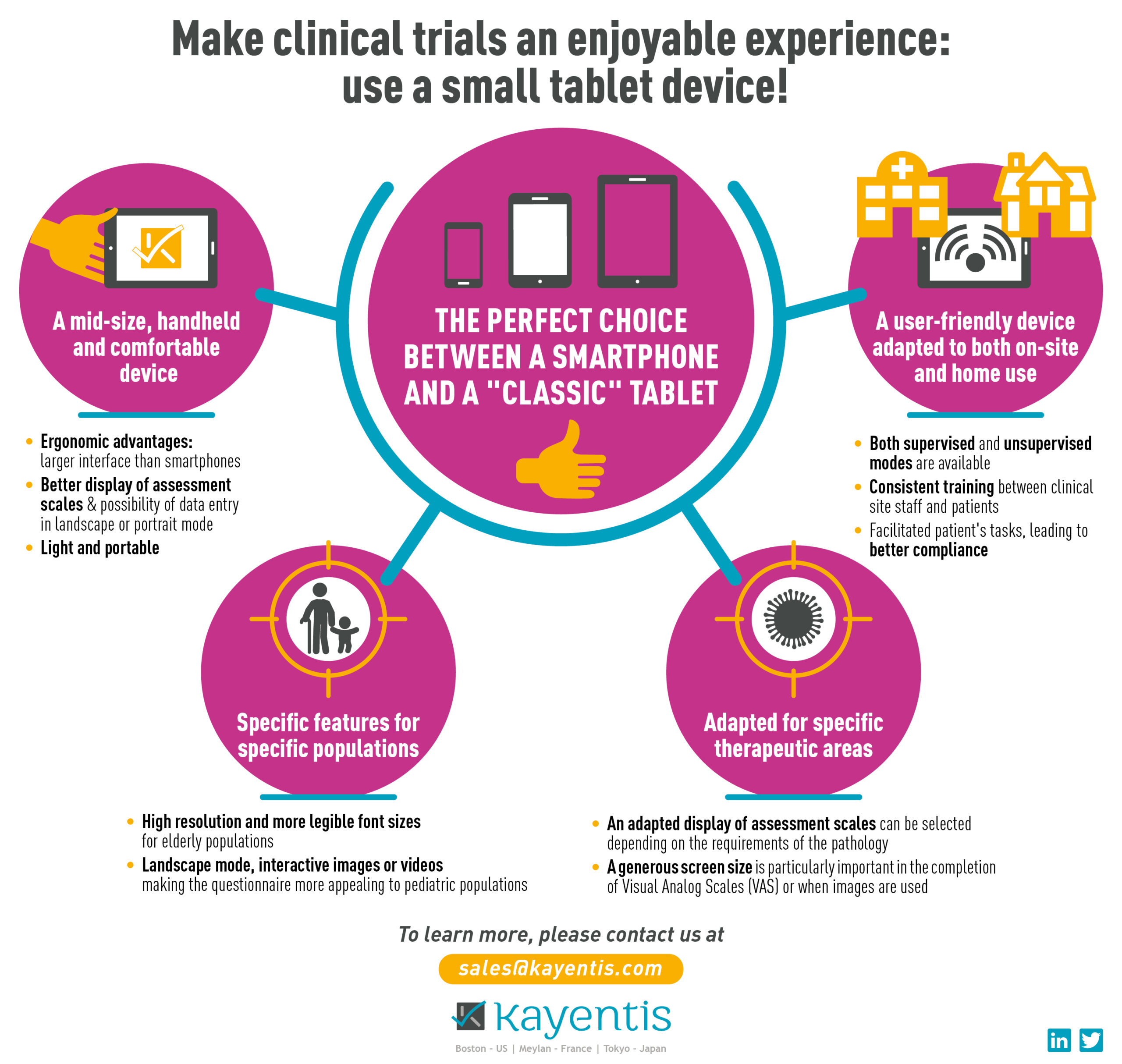
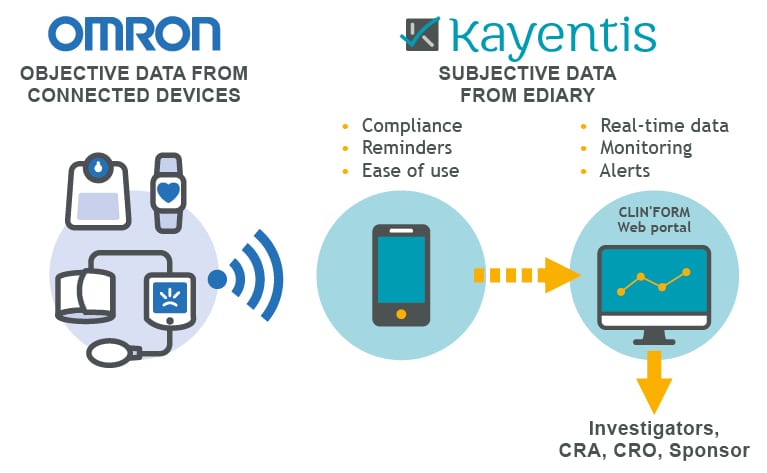
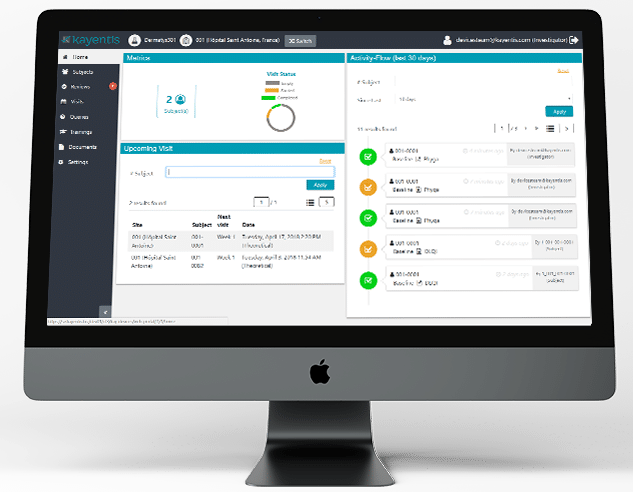
Top-tier expertise in electronic Clinical Outcome Assessment (eCOA) and Interactive Response Technology (IRT), combined in seamless integrated solution, will enable sponsors and CROs to streamline clinical trial workflows, reduce digital...