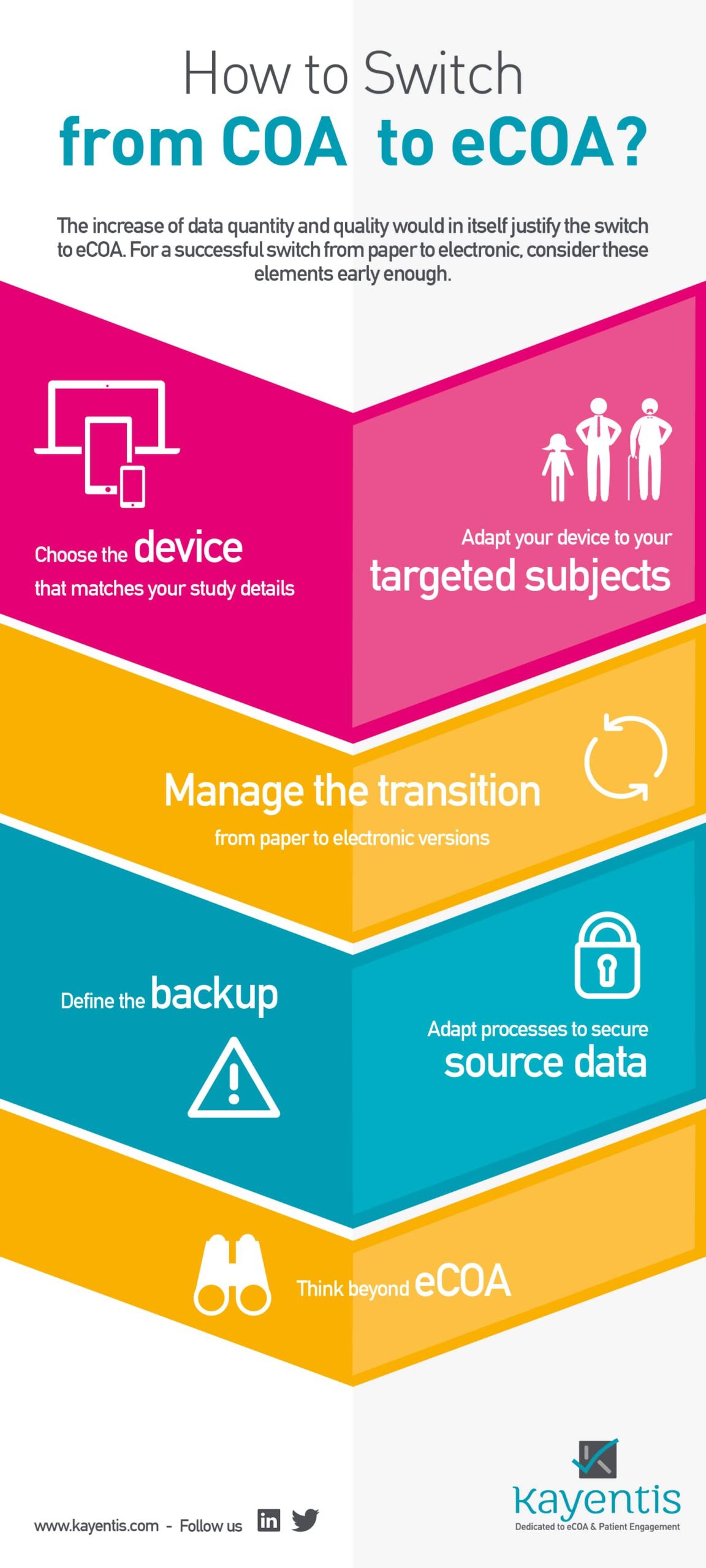
Webcast
What if eCOA could elevate your next rare disease trial?
Interview with Prof. Olivier Blin, Professor of Pharmacology, rare disease expert, and Orphan Dev coordinator Despite remarkable progress in rare disease research, 90% of rare diseases still lack an approved treatment....